Metabolic changes in the immune system modulate cell fate and function, influencing immune response outcomes. Savicell technology measures the energy changes of the immune system, which are disease-specific, and identifies the ailment.
Immune cells are the first to recognize and respond to the formation of cancer cells. When naïve lymphocytes detect cancer antigen, they undergo various differentiation processes aimed at initiating the immune response and bringing it to an optimal level of activity.
Immune cells are the first to recognize and respond to the formation of cancer cells. When naïve lymphocytes detect cancer antigen, they undergo various differentiation processes aimed at initiating the immune response and bringing it to an optimal level of activity.
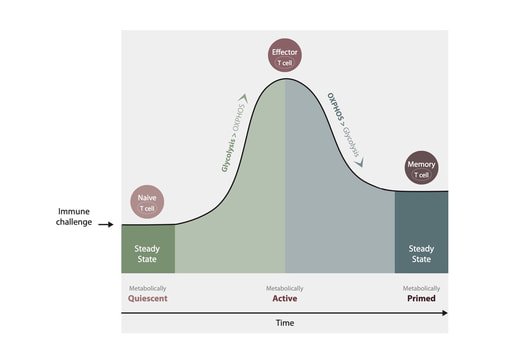
One of the most important recent discoveries is that immune system cells alter their energy generation in order to obtain an effector function. The metabolic change is called a metabolic shift, and its key purpose is command and control of the effector function, which produces and secretes cytokines and chemokines.
The immune system is composed of several groups each with a number of subgroups. Each group and subgroup has a specific energy generation profile. Example of groups are naïve cells, effector cells, regulator cells, and memory cells. The metabolic changes of the immune system cells are direct indicators of the performance of the immune system.
Immune cells use various processes to generate energy. These include oxidative phosphorylation, glycolysis, and the breakdown of proteins and nitrogenous bases. The energy generation produces changes in extracellular acidification. Lactic acid is generated in glycolysis, carbon dioxide from oxidative phosphorylation, and ammonia from the breakdown of proteins and nitrogenous bases. Acidification is measured in "open" versus "closed" (air-sealed) wells to track the accumulations of soluble versus volatile metabolic products (lactic acid versus carbon dioxide and ammonia). Savicell's tests measure these acidification (pH) changes.
Our technology exposes the immune system cells to stimulants that have been characterized specifically for the method. The process of acid production described above and the monitoring of the pH level in the cell environment allows early detection of disease. This is because the metabolic profiles of immune cells in sick people are different from those of the healthy.
Immune cells of sick people have already been activated (metabolic shift) in vivo against cancer proteins. As a result, acidification rates are different compared to the immune cells of healthy individuals that have not been exposed in vivo to cancer proteins. To measure extracellular acidification Savicell adds a pH-sensitive impermeable fluorescence probe along with the cells and stimulants to the microwell plate, which is monitored over time. Various stimulants are chosen to increase the pH signal and to more specifically characterize the disease.
Savicell ImmunoBiopsy platform uses three types of stimulants:
1. General stimulants that activate all immune system cells in a non-specific mode, and are used as positive controls for testing. Examples are para-methoxyamphetamine (PMA), lipopolysaccharides (LPS), and concanavalinA (ConA).
2. Metabolic stimulants used as substrates, whose consumption increases in activated cells, especially those undergoing increased glycolysis. Examples are glucose and L-glutamine.
3. Cancer-specific stimulants, including those by cancer type (e.g. lung). These enhance separation by specific cancer type as well as distinguishing cancer from healthy and other diseases. Examples are human epidermal growth factor receptor 2 (HER2) and New York esophageal squamous cell carcinoma 1 (NY-ESO-1).
Immune cells use various processes to generate energy. These include oxidative phosphorylation, glycolysis, and the breakdown of proteins and nitrogenous bases. The energy generation produces changes in extracellular acidification. Lactic acid is generated in glycolysis, carbon dioxide from oxidative phosphorylation, and ammonia from the breakdown of proteins and nitrogenous bases. Acidification is measured in "open" versus "closed" (air-sealed) wells to track the accumulations of soluble versus volatile metabolic products (lactic acid versus carbon dioxide and ammonia). Savicell's tests measure these acidification (pH) changes.
Our technology exposes the immune system cells to stimulants that have been characterized specifically for the method. The process of acid production described above and the monitoring of the pH level in the cell environment allows early detection of disease. This is because the metabolic profiles of immune cells in sick people are different from those of the healthy.
Immune cells of sick people have already been activated (metabolic shift) in vivo against cancer proteins. As a result, acidification rates are different compared to the immune cells of healthy individuals that have not been exposed in vivo to cancer proteins. To measure extracellular acidification Savicell adds a pH-sensitive impermeable fluorescence probe along with the cells and stimulants to the microwell plate, which is monitored over time. Various stimulants are chosen to increase the pH signal and to more specifically characterize the disease.
Savicell ImmunoBiopsy platform uses three types of stimulants:
1. General stimulants that activate all immune system cells in a non-specific mode, and are used as positive controls for testing. Examples are para-methoxyamphetamine (PMA), lipopolysaccharides (LPS), and concanavalinA (ConA).
2. Metabolic stimulants used as substrates, whose consumption increases in activated cells, especially those undergoing increased glycolysis. Examples are glucose and L-glutamine.
3. Cancer-specific stimulants, including those by cancer type (e.g. lung). These enhance separation by specific cancer type as well as distinguishing cancer from healthy and other diseases. Examples are human epidermal growth factor receptor 2 (HER2) and New York esophageal squamous cell carcinoma 1 (NY-ESO-1).
