Savicell's proprietary ImmunoBiopsy™ technology has broad application in identifying disease status in multiple diseases that include cancer and autoimmune diseases. Savicell immune platform measures metabolic changes in the immune system. These energy changes to glycolysis give the immune system the ability (effector function) to attack disease, including cancer. Diseases produce unique signatures in immune metabolic profiles that ImmunoBiopsy™ can use to distinguish between and within various ailments. It may be used to track treatment effectiveness, including immunotherapy. Our initial focus is on lung cancer.
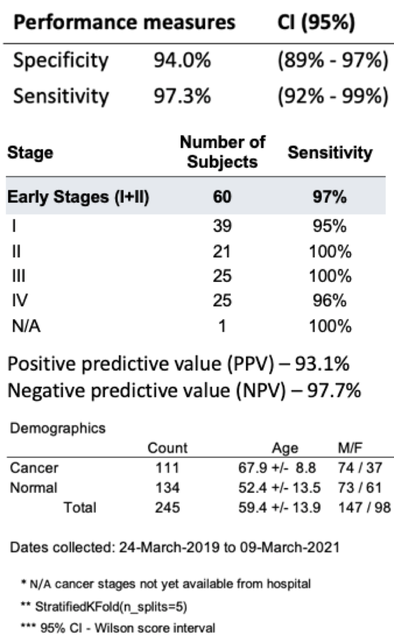
ImmunoBiopsy™ demonstrated multiple compelling, cross validated clinical studies in lung cancer. Including studies published in Cancer Immunology and Immunotherapy and Respiratory Research. The later published study published in Feb 2023 showed specificity and specificity of 94% and 97%, respectively.
Abstract
Introduction
Lung cancer remains the leading cause of death from cancer, worldwide. Developing early detection diagnostic methods, especially non-invasive methods, is a critical component to raising the overall survival rate and prognosis for lung cancer. The purpose of this study is to evaluate two protocols of a novel in vitro cellular immune response test to detect lung cancer. The test specifically quantifies the glycolysis metabolism pathway, which is a biomarker for the activation level of immune cells. It summarizes the results of two clinical trials, where each deploys a different protocol’s version of this test for the detection of lung cancer. In the later clinical trial, an improved test protocol is applied.
Method
The test platform is based on changes in the metabolic pathways of the immune cells following their activa- tion by antigenic stimuli associated with Lung cancer. Peripheral Blood Mononuclear Cells are loaded on a multiwell plate together with various lung tumor associated antigens and a fluorescent probe that exhibits a pH-dependent absorption shift. The acidification process in the extracellular fluid is monitored by a commercial fluorescence plate reader device in continuous reading for 3 h at 37 °C to document the fluorescent signal received from each well.
Results